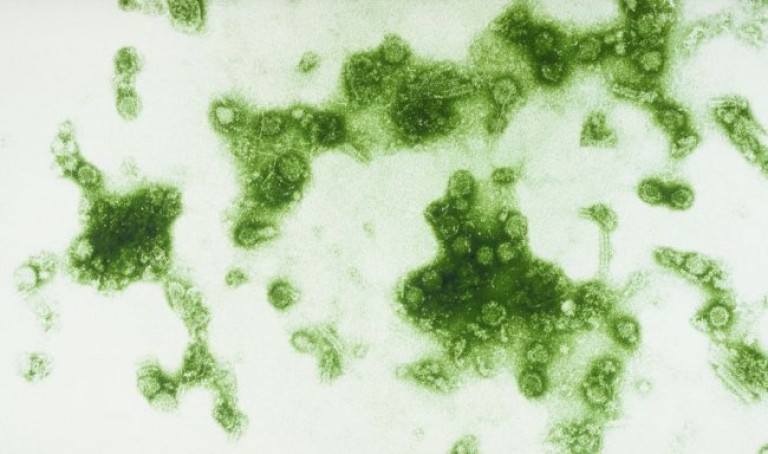
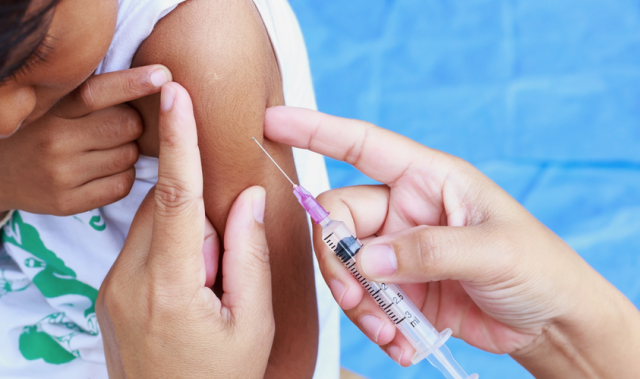
AsianScientist (Dec. 28, 2015) – Sanofi Pasteur, the vaccines division of Sanofi, has been granted marketing approval for Dengvaxia® in the Philippines, making it the first vaccine to be licensed for the prevention of dengue in Asia.
The Philippines’ Food and Drug Administration approved Dengvaxia®, a tetravalent dengue vaccine, for the prevention of disease caused by all four dengue types in individuals from 9-45 years of age living in endemic areas.
“Approval of the first dengue vaccine in Asia, which bears 70 percent of global disease burden, is a major milestone in dengue prevention and public health,” noted Mr. Olivier Charmeil, President and CEO of Sanofi Pasteur.
“Approval of Dengvaxia® in the Philippines, following closely the first approval in Mexico, is further evidence of Sanofi Pasteur’s long-standing commitment to introduce this innovative new vaccine first in countries where dengue is a major public health threat,” added Charmeil.
The dengue fever burden in Asia continues to be the highest globally with an estimated 67 million people being sickened by the dengue annually. As an urban disease, dengue attacks populations of Asia in the form of unpredictable outbreaks capable of paralyzing health care systems, negatively impacting social and economic activity. Asian endemic countries spend an estimated US$6.5 billion annually in both direct medical and indirect costs due to dengue.
Dr. May Book Montellano, President, Philippine Foundation for Vaccination, said that prevention of dengue is an urgent and growing medical priority in the Philippines.
“Vaccination is widely accepted as one of the most cost-effective ways to reduce the spread of infectious diseases like dengue. The approval of Dengvaxia®, the world’s first dengue vaccine, in the Philippines will be a critical addition to the ongoing public education and vector control efforts currently directed towards dengue prevention in our country,” said Montellano.
Sanofi Pasteur enrolled over 40,000 participants in extensive safety and clinical efficacy studies conducted mainly in endemic countries and built a dedicated vaccine production facility in France to secure adequate quality and quantity supply of the vaccine to meet endemic country demand upon introduction.
Although dengue affects people from all ages and socio-economic backgrounds, the greatest number of dengue cases worldwide occurs in the highly mobile and social segment of endemic populations that include pre-adolescents to adult ages of nine years and above.
———
Source: Sanofi Pasteur; Photo: Animal Diversity Web/James Gathany/Flickr/CC.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.