AsianScientist (Aug. 31, 2016) – Japanese scientists have shown through simulations that four phases of a substance can coexist at thermal equilibrium, where all parts are at the same temperature and pressure—a situation that seemingly goes against the laws of thermodynamics.
The findings, published in Nature Communications, not only deepen our basic understanding of phases of a substance existing in equilibrium, but may also be applied to the development of functional materials possessing phase-change properties.
We constantly come across instances where various phases of a substance—such as gas, liquid, and solid—coexist with each other. For example, water (liquid) and ice (solid) coexist in shaved ice, a popular summer treat. Furthermore, the three phases of gas, liquid, and solid in water molecules coexist at a particular temperature and pressure known as the triple point.
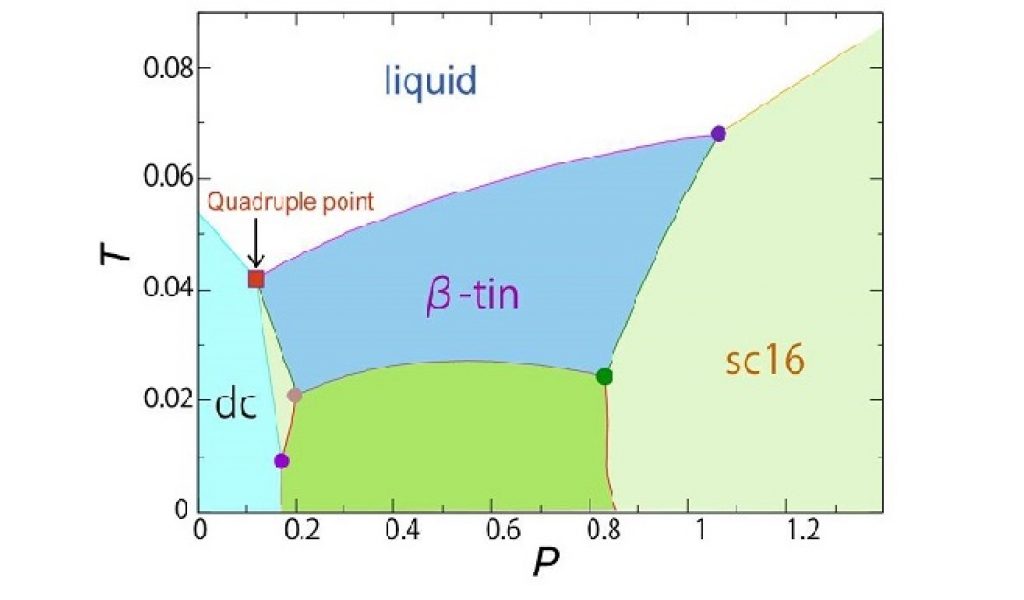
In a new study, the research group led by Professor Hajime Tanaka of the Institute of Industrial Science at the University of Tokyo has shown that four phases—three crystalline phases and one liquid phase—can coexist. These findings violate the Gibbs phase rule, which states that no more than three phases of a substance made from a single component can coexist at thermal equilibrium.
“We usually are not taught that there can be a special case apparently violating this rule. Our study shows that by changing the interaction potential of a system, which is usually material-specific, we can search such a special condition in a systematic manner,” said Tanaka.
Moreover, the group systematically determined the particular temperature and pressure of the quadruple point where the four phases coexist, and defined the phase transition behavior—switching from one phase to another—around this point.
This study may also prove useful in the development of functional phase-change materials as it demonstrates that multiple phase transformation can be induced near the quadruple point.
The article can be found at: Akahane et al. (2016) A Possible Four-phase Coexistence in a Single-component System.
———
Source: University of Tokyo.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.