AsianScientist (May. 12, 2023) –While you are reading this, photoreceptors—specialized light-sensing neurons in your retina—are busy translating light into electrical and chemical signals for the brain to assemble the image you perceive. The deterioration of photoreceptors in retinal disorders like age-related macular degeneration (AMD) and the rare inherited condition, retinitis pigmentosa, is one of the primary causes of declining vision and eventual blindness worldwide. No current treatment options are available yet that can help regenerate these essential cells. Although scientists have been making progress toward developing different therapeutic methods for these debilitating conditions, limitations in their reproducibility and feasibility have hindered their clinical application.
Recently, scientists from Singapore and Sweden reported a promising stem cell therapy approach that could potentially restore vision by replacing photoreceptors lost in patients with degenerative retinal disorders. The study was published in Molecular Therapy.
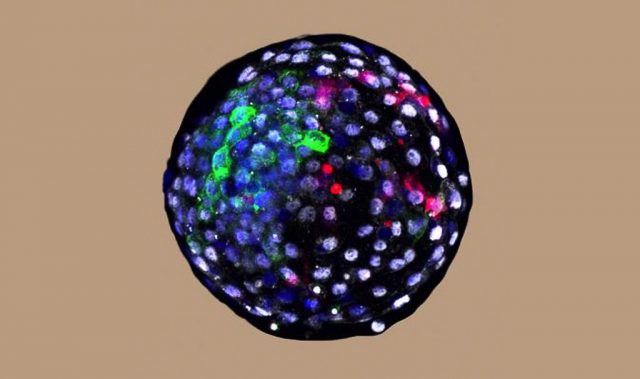
Researchers from Duke-NUS Medical School, the Singapore Eye Research Institute and the Karolinska Institute in Sweden have joined forces to meet this challenge. Together, they devised a highly-reproducible approach to transform stem cells into progenitor cells of photoreceptors, which were found to partially restore vision upon transplantation into animal models having damaged retinas.
The team grew human embryonic stem cells in the presence of a specific version or isoform of laminin—a protein that is typically enriched in the retina. In under 32 days, the stem cells differentiated into photoreceptor progenitors and began expressing markers that are characteristic of these cells.
“We hypothesize that the recapitulation of the human retina matrix by the use of laminin isoforms could help support and drive the differentiation of human embryonic stem cells into photoreceptors,” said Tay Hwee Goon, Assistant Professor from Duke-NUS’ Centre for Vision Research and lead author of the study, to Asian Scientist Magazine.
Four weeks after transplanting these stem cell-derived photoreceptors into the damaged retinas of rodents , the cells began to express markers similar to those of mature photoreceptors. Visual behavioral assessment using the water maze swimming test showed a significant improvement in vision. The team also performed an electroretinogram to demonstrate a significant recovery in the damaged retinas by measuring electrical activity in the retina in response to a light stimulus. Moreover, studies conducted over a longer period of 20 weeks revealed that the transplanted cells formed connections with nearby host cells and survived effectively for several weeks after the transplant.
In addition to these promising results, Tay and her team chose to deviate from the conventional approach to cell culture for boosting the likelihood of this therapy being approved for clinical use. Unlike common cell differentiation media that contain bovine or calf serum which can lead to variations in different batches, they used a unique growth medium made from chemically defined and animal product-free ingredients.
“The high-quality control of the cell product which results from consistent cell manufacturing is highly sought after because that would translate to more consistent test outcomes. Taken together, it may positively impact the reviewing process of the regulatory bodies for clinical approval,” said Tay.
These findings imply an exciting first step toward a potentially viable regenerative therapy that can reverse retina damage and treat declining vision caused by photoreceptor loss. The protocol of the procedure developed by Tay and her colleagues at Duke-NUS has been licensed to Alder Therapeutics, a Swedish biotech start-up. Tay shared that Alder Therapeutics will focus on quality control in the manufacturing of clinical-grade stem cell-derived photoreceptor progenitors.
“These cells will subsequently be tested for safety and efficacy extensively in pre-clinical models within these 5 years before advancing towards human clinical trials. These steps are important to shape and optimize the final cell product to be used commercially if the clinical trial is successful,” said Tay.
Source: Duke-NUS Medical School ; Image: Shutterstock
The article can be found at Photoreceptor laminin drives differentiation of human pluripotent stem cells to photoreceptor progenitors that partially restore retina function.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.