AsianScientist (Feb. 4, 2021) – Using mathematical modeling, researchers from the Korea Advanced Institute of Science and Technology (KAIST) have identified how aging and conditions like dementia and obesity cause sleep disorders. Their findings were published in the Proceedings of the National Academy of Sciences.
Whether you’re an early bird or a night owl, our sleep-wake cycles are all governed by an internal clock known as the circadian rhythm. The aptly-named PERIOD (PER) protein is one such factor that plays a crucial role in regulating the cyclical pattern of our circadian rhythms.
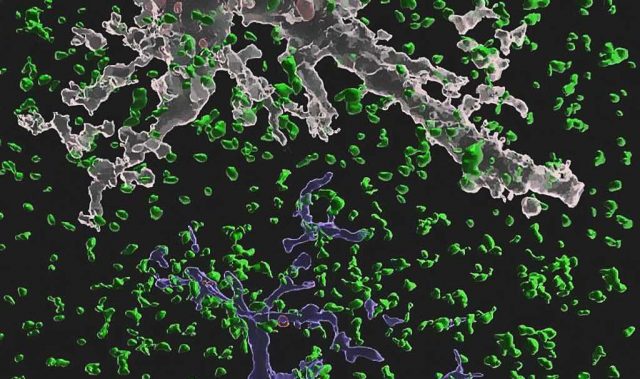
As soon as we wake up, PER starts to build up in our cytoplasm. Once a certain threshold of the protein has been reached, all of the cytoplasmic PER molecules enter the cell nucleus at once. In turn, the sudden influx of PER hinders the transcription of PER genes—reducing the amount of PER in the process. The cycle is then repeated the next day.
Given the complexity of the cell’s environment, it remains unknown how thousands of PER molecules can enter the nucleus at the same time. To solve this mystery, Professor Kim Jae Kyoung from KAIST and his team developed a mathematical model that describes the motion of PER molecules in the cytoplasm.
According to the team’s model, PER has to be sufficiently concentrated around the cell nucleus before it can be ‘switched on’ via phosphorylation and enter the nucleus collectively. These predictions were validated by in vivo experiments, which showed a sudden jump in the phosphorylation of PER rather than a gradual accumulation of phosphorylated PER over time.
The model also predicted that cytoplasmic crowding—which prevents the accumulation of PER around the nucleus—would disrupt this switch-like behavior of PER and thereby destabilize the circadian rhythm. Indeed, the researchers found that the accumulation of fat around the nucleus altered the phosphorylation of PER and disrupted the circadian rhythm. Furthermore, the circadian rhythm could be restored if PER levels were increased.
These results provide clear mechanistic insight into why circadian and sleep disorders arise in such clinical conditions as metabolic and neurodegenerative diseases and aging, in which the cytoplasm is congested.
“As a mathematician, I am excited to help enable the advancement of new treatment strategies that can improve the lives of so many patients who suffer from irregular sleep-wake cycles. Taking these findings as an opportunity, I hope to see more active interchanges of ideas and collaboration between mathematical and biological sciences,” Kim said.
The article can be found at: Beesley et al. (2020) Wake-sleep Cycles Are Severely Disrupted by Diseases Affecting Cytoplasmic Homeostasis.
———
Source: Korea Advanced Institute of Science and Technology; Photo: Shelly Liew/Asian Scientist Magazine.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.