AsianScientist (Jul. 30, 2018) – The survival of mice cloned using somatic cell nuclear transfer (SCNT) can be improved with epigenetic modifications. These findings have been published in Cell Stem Cell.
Cloned animals like Dolly the sheep are created through a process called SCNT, where the nucleus of a normal cell in the body is transplanted into a reprogrammed egg cell that has the potential to become any type of cell. If all goes well, a genetically identical clone of the donor individual is created.
In the case of Dolly, the procedure had to be carried out manually more than 277 times before the first fetus was successfully brought to term. Recent research has revealed that epigenetic factors—changes to the chemical modifications of DNA or histone proteins rather than to the genetic code itself—play a key role in hampering the proper development of the clones.
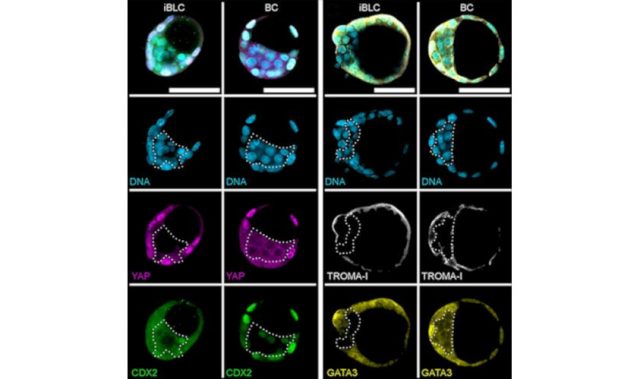
In the present study, scientists led by to Dr. Shogo Matoba of the RIKEN BioResource Research Center, Japan, performed epigenetic modification on SCNT embryos to improve the success of embryo implantation and development. The researchers combined two approaches: removing an RNA species known as Xist, followed by injecting Kdm4d messenger RNA.
Xist, which normally is only expressed from the paternal X chromosome, is expressed from both X chromosomes in SCNT, leading to problems in development after implantation. Meanwhile, Kdm4d is an enzyme that removes methyl groups from histone H3 to improve the rate of successful implantation of cloned embryos.
Hence, by getting rid of Xist and increasing levels of Kdm4d, the researchers were able to ensure that approximately 20 percent of the implanted cells were brought to term. The work also revealed that even when embryos implanted successfully, they often had abnormalities that prevented them from developing normally. Sometimes, there were abnormalities in the placenta itself.
The researchers also found that although the general level of epigenetic marks was similar in fertilized embryos and SCNT embryos, there were differences in the specific genes that were silenced through methylation. Additionally, a specific set of genes that was not expressed from the maternal chromosome in fertilized embryos was expressed from both the paternal and maternal chromosomes in SCNT embryos. These genes are known to be involved in the development of the fetus and placenta, providing a likely explanation for the failure of development after implantation.
“With this work, we have definitively shown that epigenetic modifications will need to be modulated to further improve the efficiency of SCNT. We have made good progress, and have also identified a key set of genes that, we believe, impedes the proper development of the fetus after implantation. We will continue studies to find ways to overcome this further barrier, hopefully leading to even better efficiency of cloning,” said Matoba.
The article can be found at: Matoba & Zhang (2018) Somatic Cell Nuclear Transfer Reprogramming: Mechanisms and Applications.
———
Source: RIKEN; Photo: Shutterstock.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.