AsianScientist (Dec. 2, 2014) – A system for viewing protein reactions at the molecular level has helped to shed light on the formation of fibrils associated with Alzheimer’s disease. This research has been published in Scientific Reports.
Examining the reactions between proteins is critical in the drug discovery process. For example, when a protein causing a disease (target) is identified, another protein with a strong affinity for the target will be examined as a candidate for use as an therapeutic agent. Knowing which protein bonds to which part of the target and with what bonding force is therefore very important in assessing the ability of the protein as an agent.
Researchers have previously tried to integrate a system for measuring affinity between proteins with fluorescent microscopy, however, the metal coating on the chips used prevented optical penetration.
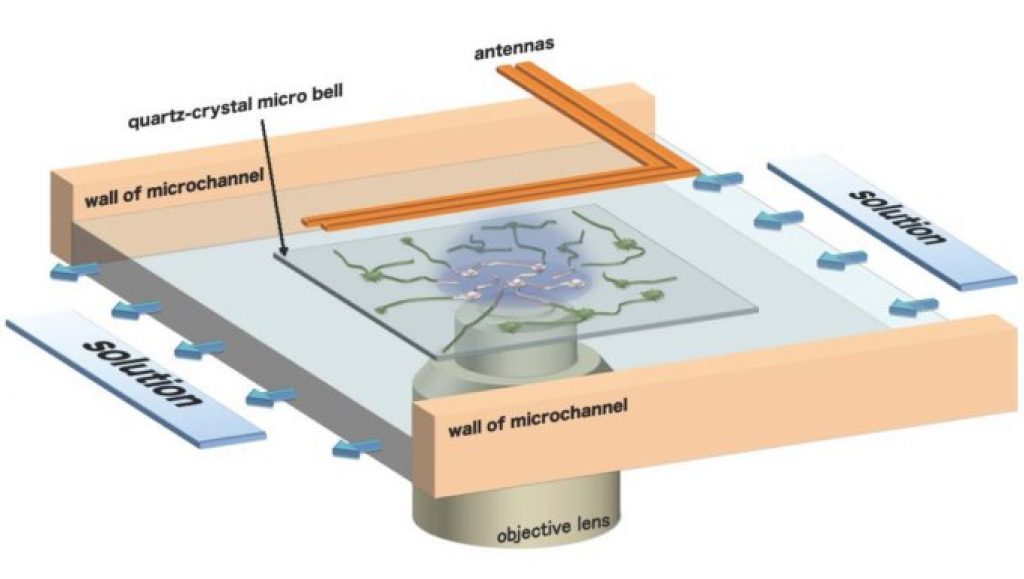
Instead of metal, a research team led by associate professor Ogi Hirotsugu and professor Goto Yuji from Osaka University have turned to quartz, developing a novel total-internal-reflection-fluorescence microscopy with a quartz-crystal microbalance (TIRFM-QCM) system. Using this system, they were able to clarify the mechanism of amyloid β (Aβ) peptide aggregation, a phenomenon associated with the pathogenesis of Alzheimer’s disease.
The key to their success was the use of a colorless and transparent quartz-crystal chip with high light permeability in protein solution. This allowed them to measure the vibrations of the quartz resonator with a pair of line antennas which generated and indirectly detected the pure-shear vibrations of the quartz resonator from the electromagnetic waves.
The researchers used the TIRFM-QCM system to monitor the deposition reaction of Aβ peptides on immobilized seeds of Aβ. They observed a fibril-elongation rate two orders of magnitude higher than previously reported values, indicating ultrafast transition of oligomers into fibrils.
Other than explaining the development of Alzheimer’s disease, the technique developed in this study could also be used to study other protein reactions at the molecular scale, making it a particularly useful tool for drug discovery.
The article can be found at: Ogi et al. (2014) Ultrafast Propagation of β-amyloid Fibrils in Oligomeric Cloud.
——–
Source: Osaka University.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.












