AsianScientist (Dec. 12, 2011) – A research team at the Chinese University of Hong Kong (CUHK) has released study results showing how Helicobacter pylori, a bacterium that causes more than half of peptic ulcers worldwide and that has been implicated in stomach cancer, has managed for ages to turn the acidic environment of the human gut into a neutral one where it can thrive.
In the Journal of Biological Chemistry’s latest “Paper of the Week,” the scientists say the information they have obtained about the pathogen’s clever employment of acid neutralizers may inform those who are designing new drugs to blunt H. pylori‘s effects across the globe.
H. pylori are the only bacteria known to thrive in the human stomach. The damage the bacteria do to the mucous coating of the gut allows stomach acid to eat away at the sensitive organ lining, causing ulcers.
Existing antibiotics can cure 80 to 90 percent of ulcers caused by the pathogen. However, H. pylori over the years have become increasingly resistant to antibiotics. Some experts have attributed that resistance to the fact that doctors are quick to prescribe antibiotics to kill it even when patients show no symptoms.
“It’s the pathogen’s ability to persist within the acid bath in the human stomach that has made it such a successful, albeit harmful, vector, said lead author Ivan Fong from CUHK.
“The key is its use of an enzyme called urease to neutralize gastric acid,” he explained.
H. pylori produce urease to spur the breakdown of urea, a naturally occurring chemical in the body, so that urea can release ammonia and make the gut an environment in which the pathogens can thrive. But, unlike most other enzymes, urease needs two nickel ions to get activated, allowing for the enzyme to mature.
“As the survival of H. pylori depends on active urease, this is a life-or-death issue for the pathogen to ensure nickel ions are delivered to the urease,” said senior author Prof. Kam-Bo Wong from the CUHK.
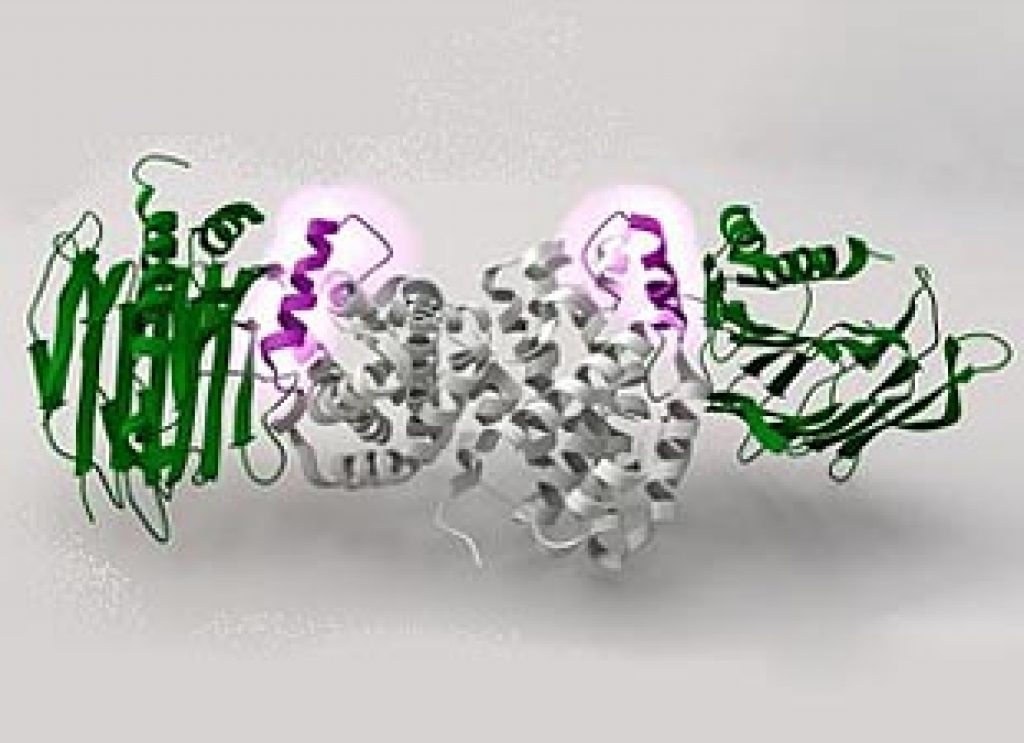
It’s not entirely clear how H. pylori make sure that urease can mature and then neutralize the surrounding acid. But Wong’s team focused on four proteins that they suspect are helpers: UreE, UreF, UreG, and UreH.
Using X-ray crystallography to visualize proteins with atomic resolution, the team took snapshots of UreF and UreH. What they saw was that UreH morphs the shape of UreF to enable UreF to recruit a third player, UreG, to form the UreF-UreH-UreG complex.
In other words, the three proteins hook up to collectively deliver nickel ions to the right place on urease. Once the nickel ions are in place, they serve like a flint to ignite the breakdown of urea into ammonia, which then neutralizes the stomach acids.
Importantly, the team also discovered that disrupting the formation of the crafty UreF-UreH-UreG complex does, in fact, inhibit the synthesis of active urease. They hope that the information they’ve obtained about the molecular structures of UreF and UreH will help in the design of drugs that will disable the works of this molecular machine.
“As active urease is the key to survival of H. pylori, designing drugs that target this complex may well be a viable strategy to eradicate the pathogen,” said Prof. Wong.
The article can be found at: Fong YH et al. (2011) Assembly of the preactivation complex for urease maturation in Helicobacter pylori: Crystal Structure of the UreF/UreH complex .
——
Source: ASBMB.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.












