AsianScientist (Aug. 11, 2011) – Guided Therapeutics, Inc., which develops rapid and painless testing platforms using biophotonics for the early detection of disease, announced yesterday that it has begun human testing of its light-based detection technology for Barrett’s Esophagus, a precursor for esophageal cancer.
The technology is being jointly developed with Konica Minolta Opto of Japan.
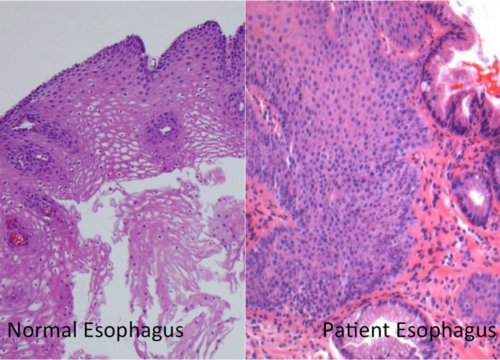
The biophotonic disease detection platform, which consists of a base unit and single-patient-use calibration disposable, scans tissue with light to identify changes in esophageal tissue called Barrett’s Esophagus, a precursor for esophageal cancer. Barrett’s esophagus is believed to be caused by excessive acid reflux.
Unlike traditional tests, this test does not require laboratory analysis or a tissue sample, is designed to provide results immediately and eliminate costly unnecessary testing.
The study also tests various hardware and procedural configurations and is designed to determine a way forward for product development and further clinical trials.
The feasibility study is expected to enroll about 40 subjects and be completed before the end of 2011 at two Atlanta-area clinics.
“The start of the human clinical feasibility study is the most significant milestone to date in the development of our pipeline Barrett’s Esophagus detection and monitoring product with partner Konica Minolta,” said Mark L. Faupel, CEO and president of Guided Therapeutics, Inc.
“This comes on the heels of announcing our Asian regional partnership with Konica Minolta for our LuViva Advanced Cervical Scan for distribution and screening clinical trials.”
According to the World Health Organization (WHO), esophageal cancer ranks just below cervical cancer in newly diagnosed cases. New cases of esophageal cancer are estimated at 410,000 worldwide, with more than 16,000 new cases a year and more than 14,000 deaths in the US alone.
Another partnership with Konica Minolta is for Guided Therapeutics’s LuViva™ Advanced Cervical Scan, a non-invasive device used to detect cervical disease instantly and at the point of care. In a multi-center clinical trial, the technology was able to detect cervical cancer in women at risk up to two years earlier than conventional modalities, according to the company.
——
Source: Guided Therapeutics, Inc.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.