AsianScientist (June 24, 2011) – Scientists from Singapore and the United States have made the striking discovery that Barrett’s esophagus, the precursor of the more deadly esophageal cancer, can form without mutations in a matter of a few days.
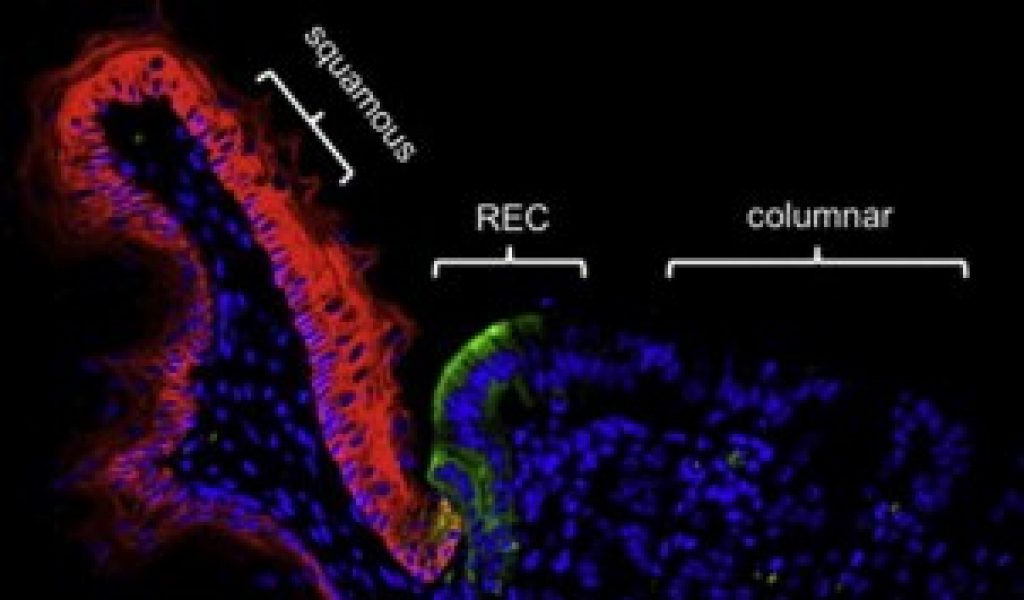
In their study, published on 24th June in Cell, the researchers traced the origins of Barrett’s esophagus, a condition where intestinal-like cells appear in the esophagus, to a very small group of embryonic cells that exist in all adults at the junction of the esophagus and the stomach.
In most people, these embryonic cells are restricted to that junction. However, if the esophagus becomes damaged by acid reflux, these embryonic cells rapidly grow to exploit this opportunity and form the unusual, intestine-like tissue of Barrett’s esophagus.
The new finding may explain why patients with gastric reflux disease often develop Barrett’s esophagus and have a higher risk of developing esophageal cancer. Esophageal cancers are difficult to treat and, together with gastric adenocarcinomas, kill more than a million people each year.
The prevailing theory has been that the abnormal cells seen in Barrett’s esophagus arise as the normal squamous stem cells “transcommit” to a new intestine-like fate as a response to acid-reflux.
To prove that Barrett’s esophagus arises from leftover embryonic cells, and not squamous stem cells, the researchers used a mouse model of chronic acid-reflux disease which lacks a gene called p63 that is required for the self-renewal of stem cells in all stratified epithelial tissues. Because of their genetic defect, the mice are born without the squamous epithelium that normally lines the esophagus.
Even without the squamous stem cells, the animals showed a vast expanse of intestine-like cells in their esophaguses with gene expression profiles very similar to those seen in Barrett’s.
The speedy development of Barrett’s-like metaplasia in the mice suggested that the precancerous condition wasn’t related to the slow accumulation of mutations. Their findings also argue against the idea that the normal stem cells were undergoing a change of fate.
“The metaplasia developed very quickly, in a matter of days,” said Dr. Wa Xian of the Institute of Medical Biology in Singapore, one of the leaders of the study. “This was shocking to us as we generally consider cancer precursors taking multiple genetic ‘hits’ and years to develop.”
The researchers also generated mouse models in which the esophageal tissue could be damaged at precise times, revealing that this damage triggered a rapid mobilization of embryonic cells that would otherwise be resting. Those cells took up the newly freed space in a process that might mimic the evolution of Barrett’s.
Again, the speed with which those cells were activated seemed to rule out mutations as an explanation in favor of competition between normal cells and the minority embryonic population.
The new findings suggest that rather than treating Barrett’s esophagus before it progresses to aggressive cancer, it may be more effective to go after the precursor cells instead.
“A lot of cancers you can do little about, and new drugs are approved based on their ability to extend life by one or two months,” said the senior author of this study, Prof. Frank McKeon of the Genome Institute of Singapore and Harvard Medical School. “Focus on the precursors of cancer may be our best hope for medicine. Here, we are looking at a precursor of a cancer precursor that is present in all of us.”
“It’s not clear that the embryonic stem cell precursors have any real purpose,” said Xian. “Methods to rid the body of those cells may therefore be the easiest and most cost-effective way to stop the disease before it even starts, particularly for those at the greatest risk.”
Whether other cancers arise by the competitive mechanism proposed for Barrett’s is unclear although McKeon and Xian say that they suspect that an additional subset of cancers, especially those linked to inflammation and tissue damage might arise from precursors derived in a manner similar to Barrett’s. If so, they anticipate rapid progress into a group of particularly aggressive cancers that typically outwit the best treatments currently available.
The article can be found at: Wang et al. (2011) Residual Embryonic Cells as Precursors of a Barrett’s-like Metaplasia.
——
Source: Elisabeth (Lisa) Lyons at Cell Press and Genome Institute of Singapore.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.












