AsianScientist (Apr. 21, 2016) – An international team of researchers at the National Institute for Physiological Sciences in Japan has identified a sequence of events in the brain that contribute to sustained mechanical allodynia, which is pain that is greatly amplified by even the slightest sensations, such as light, touch, or warmth. The findings were reported in The Journal of Clinical Investigation.
When we are in pain, we reach for painkillers as we try to ‘turn off’ the pain. Unfortunately, this does not always work, and this problem has prompted researchers around the world to seek the ‘main switch’ that can promptly and effectively conduct this switching off.
Pain comes in many forms; one is allodynia, for which current treatments are limited. The spinal cord and glial cells, which provide support and insulation between neurons and are the most abundant cell types in the central nervous system, are thought to play a crucial role in allodynia. However, the underlying cellular mechanisms of their involvement remained unsolved, until now. Notably, the S1 cortex, the region of the brain under study, is not directly affected by spinal cord injury.
“Direct manipulation of the S1 cortex has been shown to relieve neuropathic pain in humans and animals. This suggests the S1 cortex might act as a sort of central processing unit within the brain networks that mediate and/or sustain chronic neuropathic pain,” first author, postdoctoral fellow Dr. Kim Sun Kwang explained.
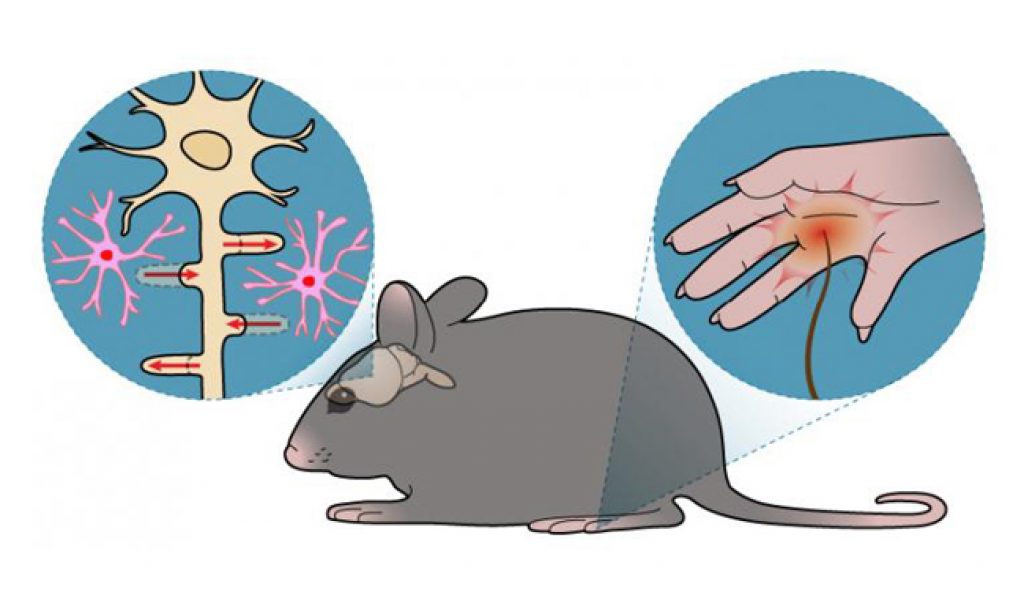
“We hypothesized that S1 astrocytes, a type of glial cell, may show functional changes following peripheral nerve injury, resulting in mechanical allodynia.”
The researchers tested the hypothesis by conducting a series of experiments in mice with a leg nerve injury. Thin filaments were applied to the underside of a back paw, and the paw response to each application was measured. At the same time, live calcium ion imaging was used to track the corresponding activities of astrocytes in the S1 cortex.
They found that specific signaling pathways, including one that allowed an influx of calcium into the astrocytes, were activated only during the first week after injury and correlated spine turnover, and that blocking the pathways suppressed mechanical allodynia. Conversely, activating them resulted in long-lasting allodynia, even when there was no peripheral injury.
The results indicate that this ‘reawakening’ of S1 astrocytes is a key trigger for S1 circuit rewiring, and that it contributes to neuropathic mechanical allodynia.
“By revealing some of the underlying mechanisms, our study suggests that cortical changes may move beyond their utility as just diagnostic tools and serve as potential targets for therapeutics,” corresponding author, Professor Junichi Nabekura said.
“Appreciation of these astrocyte-mediated changes in cortical synaptic connections requires a paradigm shift in our understanding of neuropathic pain pathophysiology; one that may result in novel therapeutic strategies for treating the debilitating effects of allodynia.”
The article can be found at: Kim et al. (2016) Cortical Astrocytes Rewire Somatosensory Cortical Circuits for Peripheral Neuropathic Pain.
———
Source: National Institutes of Natural Sciences.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.