AsianScientist (Nov. 20, 2015) – Researchers at the Tokyo Institute of Technology have developed a thin film system that can be reversibly converted from superconductor to insulator simply by changing the electric potential. Their results, published in Scientific Reports, demonstrate the possibilities of using lithium ion electrochemical reactions to control superconductivity.
“Controlling such exotic properties by applying an external field is one of the biggest continuous challenges in condensed matter physics,” said Assistant Professor Kohei Yoshimatsu of the Department of Applied Chemistry.
Observations of a transition between a superconducting and insulating state in certain two-dimensional materials has stimulated debate within the research community as to what might be the mechanisms operating in such a transition. In addition ‘high-temperature’ superconductivity, as observed in certain transition metal oxides, has prompted an interest in future applications using superconducting properties without the need for extreme cryogenic refrigeration.
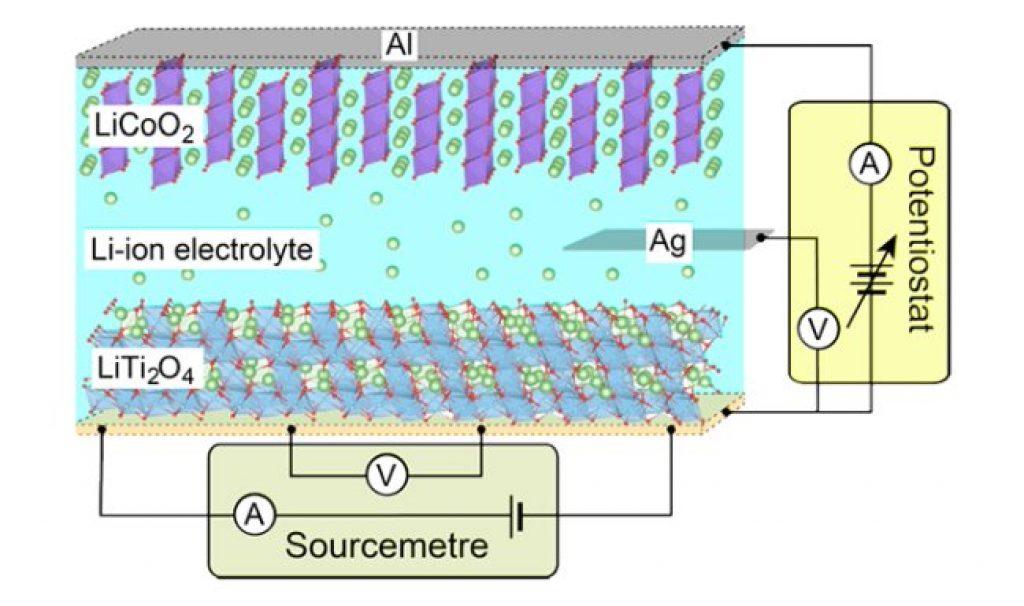
Recent work showed that electrochemically adjusting the lithium ion content in superconducting lithium titanate (LiTi2O4) can adjust the critical temperature at which the resistance drops to absolute zero. However in these experiments there was no observation of a superconducting-to-insulating state transition. Instead different phases formed and separated.
Yoshimatsu and his colleagues applied an electrochemical potential to thin films of LiTi2O4 in a set up resembling a lithium-ion battery. When the potential is applied lithium ions intercalate into LiTi2O4, ‘overdoping’ the material.
Thin films have a larger surface area and less volume than the bulk LiTi2O4 studied in previous experiments, and the researchers were able to observe a transition to an insulating material in the over-doped material. Furthermore by reversing the potential, deintercalation took place and the thin film resumed superconducting behavior.
“This complete reversible transition originates in the difference in potentials required for deintercalation of initially contained and electrochemically intercalated lithium ions,” explain the researchers.
The researchers suggest the results may lead to a better understanding of the electronic phase of thin layers.
The article can be found at: Yoshimatsu et al. (2015) Reversible Superconductor-insulator Transition in LiTi2O4 Induced by Li-ion Electrochemical Reaction.
———
Source: Tokyo Institute of Technology.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.












