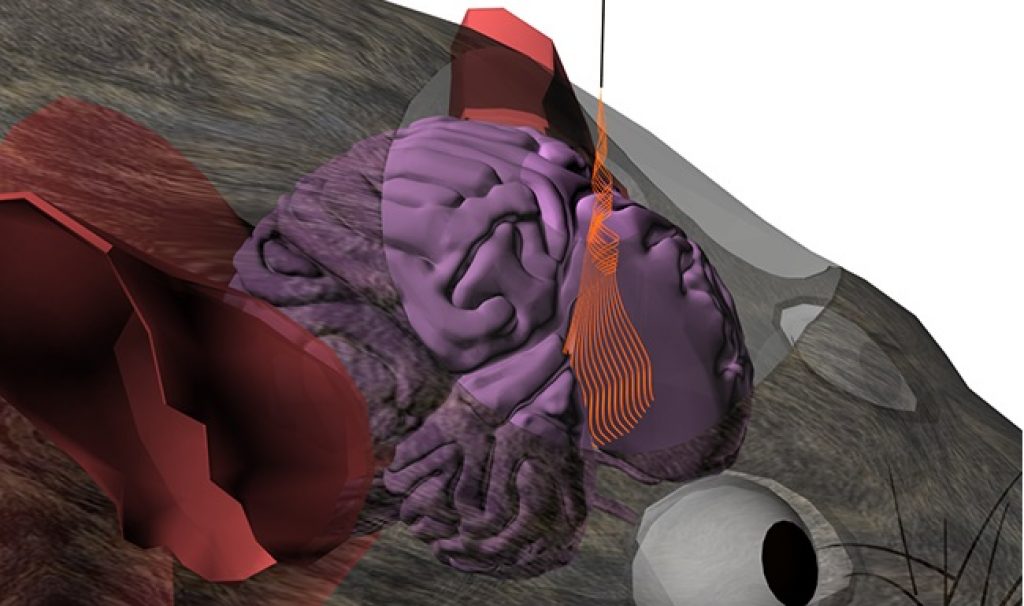
AsianScientist (Jun. 9, 2015) – In a paper published in Nature Nanotechnology, researchers describe ultra-flexible mesh electronics which can be directly injected into the brains of mice. Once there, the injected electronics integrated with brain tissue without causing chronic inflammation, suggesting that they could be used as minimally invasive detection or stimulation devices.
Developing flexible electronics that can be worn on the skin is an area of intense research. Now, a team of researchers led by Professor Charles Lieber at Harvard University have taken the concept one step further with their development of an electronic mesh than can be injected into 3D soft materials. The research was done in collaboration with Professor Fang Ying from the National Center for Nanoscience and Technology in Beijing.
Current medical implants such as pacemakers are relatively large, requiring surgery that causes discomfort to the patient and may result in serious complications. In contrast, injections are routine medical procedures.
Having developed a sub-micron thick, large-area macroporous mesh electronics in previous research, Professor Lieber and his group wanted to investigate if their mesh—which is approximately a million times more flexible than traditional flexible electronics—could be applied to living tissue and other soft materials.
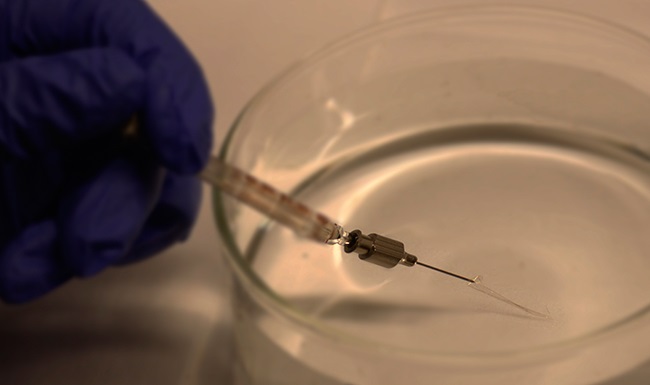
“Since no one had done this before, the fact that you could do such an injection—treating electronics as something like a reagent or drug—without damaging the circuitry was really cool!” Lieber told Asian Scientist Magazine.
Firstly, Lieber and his team showed that the flexible mesh electronics retained more than 90 percent of their functionality even after being passed through the tiny bore of a needle. Imaging and simulations showed that differences in stiffness between the transverse and longitudinal planes of the mesh allowed it to ‘roll up’ during injection, thereby preserving its structure.
When coupled with piezoelectric strain sensors and injected into the brains of mice, the electronic mesh was able to monitor brain activity. Importantly, the injecteable electronic mesh did not provoke a chronic immune response or scar tissue formation, as is commonly seen with rigid implantable devices.
“Another important finding is that after injecting electronics into a specific region of the brain, the electronics can provoke the regeneration and migration of neural cells into the macroporous electronics–that is, the mesh electronics behaved as an excellent tissue scaffold consistent with our earlier in vitro tissue engineering work,” Lieber said.
The researchers also showed that the mesh electronics can record the brain activities ranging from a single neuron action potential to local field potentials from groups of neurons in a manner that is more stable due to the absence of chronic damage. Furthermore, the injectable mesh electronics could also be used to stimulate or modulate neurons in a highly targeted yet minimally invasive manner.

Lieber and colleagues hope to demonstrate the long-term stability of their injectable mesh electronics, particularly in deep brain regions relevant to Parkinson’s disease. They are currently conducting studies on the use of injectable mesh electronics in the retina of mice and are also looking into creating multi-functional devices that can fully integrate with neural networks, blurring the distinction between the two.
In particular, due to the low-immunogenicity of the injectable mesh electronics, they foresee many applications in regenerative medicine.
“We believe our injectable mesh electronics are good candidates for cellular therapy by co-injection of mesh electronics with stem cells, for example, in treatments of stroke and other neurodegenerative disease. Our electronics can then give a powerful new means for simultaneously supporting the development of the stem cells, as well as monitoring and stimulating neural activity and connections,” Lieber said.
The article can be found at: Liu et al. (2015) Syringe-injectable electronics.
———
Copyright: Asian Scientist Magazine; Photo: Lieber Research Group/Harvard University.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.












