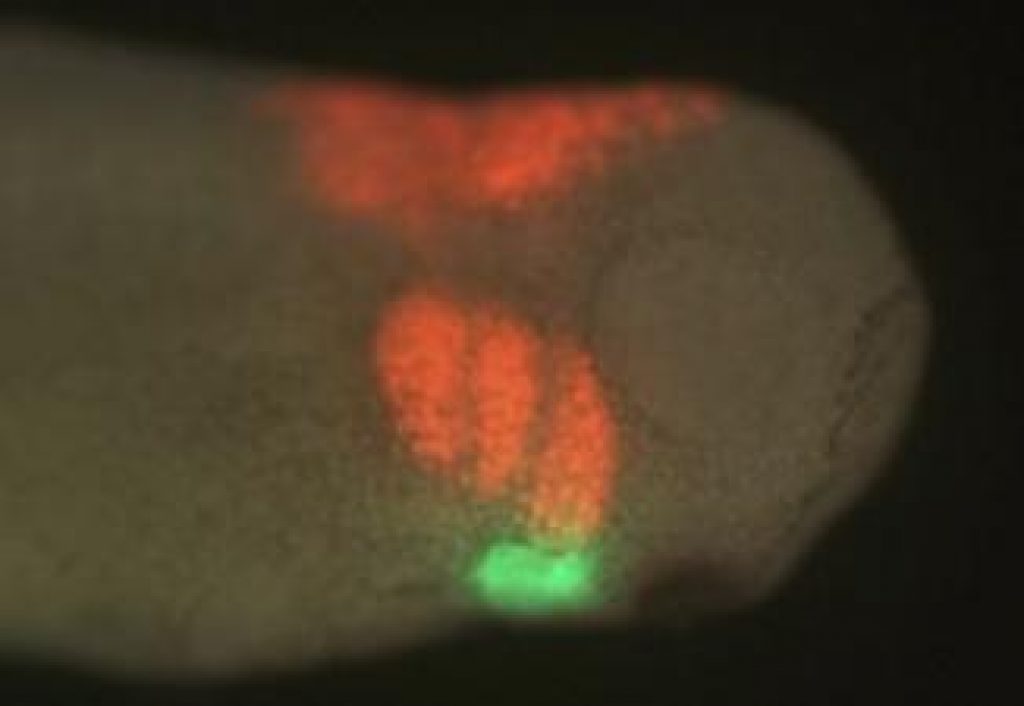
AsianScientist (Apr. 24, 2011) – Using fluorescently-dyed cells migrating to the heart, scientists have teased apart important steps during embryonic development, where cells migrate to their eventual location in the adult body plan and begin to differentiate into specific cell types.
In this week’s edition of the journal Development, Jean-Pierre Saint-Jeannet from University of Pennsylvania, USA and Young-Hoon Lee of Chonbuk National University, South Korea mapped the embryonic region that becomes the part of the heart that separates the outgoing blood in frogs.
Xenopus, a genus of frog, is a commonly used model organism for developmental studies, and is a particularly interesting for this kind of research because amphibians have a single ventricle and the outflow tract septum is incomplete.
In higher vertebrates such as chickens and mice, the cardiac neural crest provides the needed separation for both circulations at the level of the outflow tract, remodeling one vessel into two.
In fish, where there is no separation at all between the two circulations, the cardiac neural crest contributes to all regions of the heart.
“In the frog, we were expecting to find something that was in between fish and higher vertebrates, but that’s not the case at all,” said Saint-Jeannet. “It turns out that cardiac neural crest cells do not contribute to the outflow tract septum, they stop their migration before entering the outflow tract. The blood separation comes from an entirely different part of the embryo, known as the ‘second heart field’.”
In addition, as compared to other models, the migration of the cardiac neural crest in amphibians has been dramatically changed through evolution, Saint-Jeannet said.
To determine where the neural crest cells migrated during development, the researchers labeled the cardiac neural crest cells in one embryo with a fluorescent dye, grafted them onto another embryo that was unlabeled, then followed the path those marked cells took under a microscope to see where those cells ended up in the developing heart.
Knowing these paths and the biological signals that govern them could have implications in treating developmental conditions found in humans, such as DiGeorge Syndrome, the abnormal deployment of the cardiac neural crest.
DiGeorge syndrome is present in about 1 in 4,000 live births, and often requires cardiac surgery to correct. Among other developmental problems, these patients have an incomplete blood separation at the level of the outflow tract, because the cardiac neural crest does not migrate and differentiate at the proper location.
——
Source: University of Pennsylvania.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.












