AsianScientist (Nov. 3, 2020) – While diamonds are highly regarded as the hardest known material in the world, they’ve historically been considered as poor conductors of electricity—at least, until now. When deformed to strain, diamonds can conduct electricity like metals, predicted an international team of researchers from Singapore, US and Russia. Their findings were published in the Proceedings of the National Academy of Sciences.
Typically, diamonds are good electrical insulators due to their ultrawide bandgap of 5.6 electron volts. This means that a large amount of energy is needed to excite electrons in diamonds before they can carry an electric current. The smaller the bandgap, the easier it is for a current to flow.
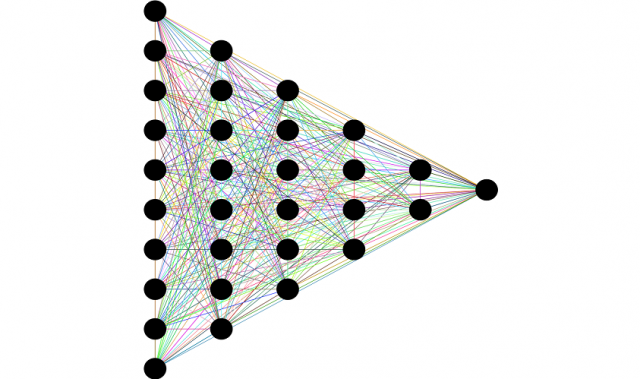
Using computer simulations, scientists co-led by Nanyang Technological University President Subra Suresh have showed that straining nanoscale diamond needles could make them behave like metals. The simulated diamonds were predicted to have altered electrical properties, including the ability to conduct electricity at room temperature and pressure, providing early proof-of-concept that deforming diamonds could narrow the bandgap.
As the amount of strain on the diamond nanoneedle increased, its predicted bandgap decreased—indicating greater electrical conductivity. In fact, the bandgap completely disappeared near the maximum amount of strain that the needle could withstand before fracture. The team also found that imparting metal-like conductivity to the diamond nanoneedle could be achieved without triggering the diamond’s transformation into graphite, the soft material found in pencils.
The simulation results were then used to train machine learning algorithms to identify conditions for optimal electrical conductivity of the nanoscale diamond in various geometrical configurations.
“The ability to engineer and design electrical conductivity in diamond without changing its chemical composition and stability offers unprecedented flexibility to custom-design its functions,” said Suresh. “The methods demonstrated in this work could be applied to a broad range of other semi-conductor materials of technological interest in mechanical, microelectronics, biomedical, energy and photonics applications, through strain engineering.”
While still at an early stage, the team’s research shows promise for expanding the application of diamonds in electronics. For instance, scientists could engineer diamond to have a bandgap comparable to silicon, widely used in semi-conductors, or even gallium nitride, which is found in light-emitting diodes. These engineered diamonds could also be used in everyday machines like electrical appliances or cars, as well as smart grids and quantum sensors.
The article can be found at: Shi et al. (2020) Metallization of Diamond.
———
Source: Nanyang Technological University; Photo: Shutterstock.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.