AsianScientist (Jun. 16, 2015) – Researchers have uncovered the molecular pathway which downregulates the transcription factor FOXP3 in response to inflammatory cues. Their findings have been published in the Proceedings of the National Academy of Sciences.
Activity of the forkhead family transcription factor FOXP3 determines the immune function of FOXP3+ Regulatory T cells (Tregs). Upon infection or other inflammatory conditions, FOXP3+Tregs may suppress effector immune cell responses leading to the failure of clearing infection or autoimmunity. FOXP3+Tregs may also help to limit collateral tissue damage during heightened inflammation.
How Treg cells sense inflammation and shut down its immune suppressive activity to allow immune activation to occur remains largely unknown. Furthering understanding of the regulation of FOXP3 stability and its dynamic ensemble of enzymatic co-factors in Tregs can thus provide therapeutic clues on how to control major inflammatory diseases including autoimmunity and allergic diseases.
In the present study, Ms. Gao Yayi and Mr. Tang Jiayou, supervised by Professor Li Bin at Institut Pasteur of Shanghai (IPS) and Professor Zheng Songguo at Penn State University Hershey College of Medicine, identified the adaptor protein Deleted in breast cancer 1 (DBC1) as a key regulator of FOXP3 stability at the protein level.
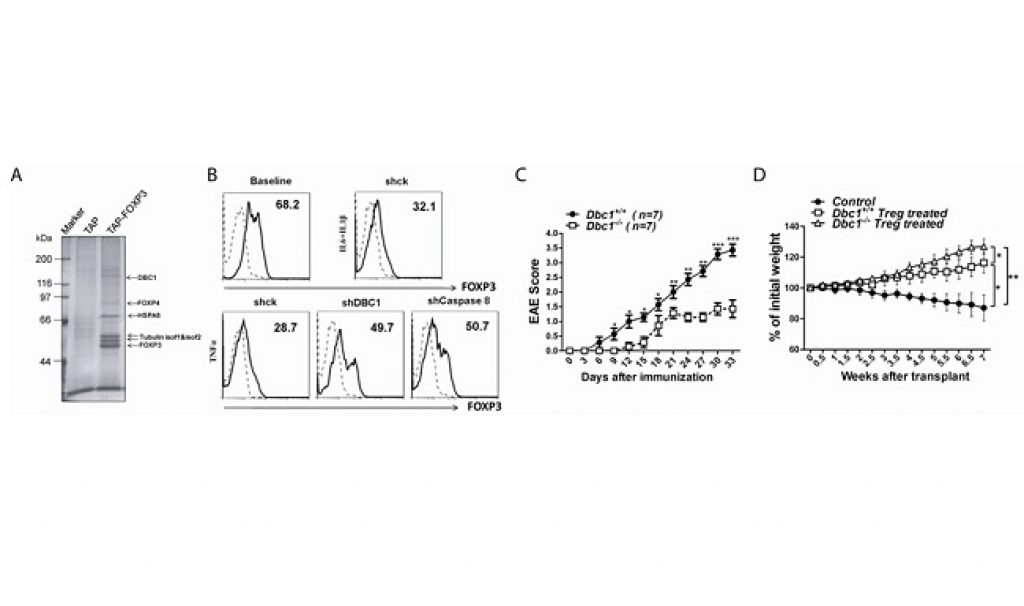
Gao purified FOXP3 transcriptional complex in FOXP3 stable expressing T cells by tandem affinity purification and found that DBC1 is a previously unidentified sub-unit of FOXP3 complex. The researchers observed that the linker region between the leucine zipper motif and the forkhead domain of FOXP3 interacts with the N-terminal 200 amino acid region of DBC1. Knockdown of DBC1 in FOXP3+Treg cells prevented caspase 8-mediated degradation of FOXP3 under inflammatory cytokine stimulation.
Moreover, Tang demonstrated that more Treg cells existed in Dbc1-/- mice and Dbc1-/- FOXP3+Treg cells are more functionally potent compared to Treg cells in wild type mice. This research proposed that DBC1 may act as a negative regulator of FOXP3 by attenuating FOXP3 protein stability and activity under inflammation, which could represent a novel molecular pathway for therapeutically modulating FOXP3+Treg function.
The article can be found at: Gao et al. (2015) Inflammation Negatively Regulates FOXP3 And Regulatory T-cell Function Via DBC1.
———
Source: Chinese Academy of Sciences.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.