AsianScientist (Mar. 15, 2011) — The conceptual and technical breakthroughs by Shinya Yamanaka (山中 伸弥, Yamanaka Shin’ya) have launched a new era in regenerative medicine and drug delivery.
That it may be possible to reverse the clock to fashion cells that possess all possible fates from those that have arrived at a single one was previously unimaginable, but proven to be possible by this orthopedic surgeon.
His training years

Shinya Yamanaka (1962, Osaka) received his M.D. from Kobe University in 1987, before completing his residency in orthopedic surgery at Osaka National Hospital in 1993. After a postdoctoral fellowship at the Gladstone Institute of Cardiovascular Disease in San Francisco, he became an assistant professor at Osaka City University Medical School, followed by the Nara Institute of Science and Technology.
Dr. Yamanaka is currently Director of the Center for iPS Cell Research and Application (CiRA) at Kyoto University (Japan), which was recently established to take a leading role in the development of iPS cell technology. He is also a professor in the Institute for Frontier Medical Sciences at the same institution. Dr. Yamanaka also currently serves as a senior investigator at the Gladstone Institute of Cardiovascular Disease in San Francisco and Professor of Anatomy at the University of California in San Francisco.
An alternative to embryonic stem cells
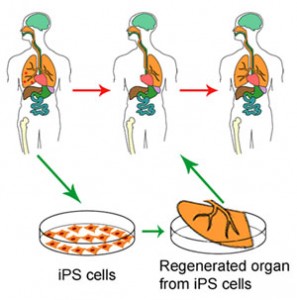
Originally an orthopedic surgeon, Dr. Yamanaka made his breakthrough discovery in 2006 when he succeeded in generating induced pluripotent stem cells or iPS cells that had the ability to differentiate into virtually any kind of tissue. These iPS cells are obtained from already specialized adult cells that are reprogrammed to pluripotency, such that they are once again able to differentiate into other tissues.
Dr. Yamanaka’s breakthrough represented a paradigm shift in developmental biology. Until then, scientists believed that pluripotency could only be achieved with embryonic stem (ES) cells – cells which hold future promise for treating injury and disease because of their ability to grow into virtually any type of differentiated cell.
ES cells, however, are not without ethical concerns, since traditional technologies derive stem cells from human embryos which are destroyed in the process. There is also the risk of immunological rejection of transplanted cells, since transplanted embryonic stem (ES) cells are not the patient’s own cells.
Dr. Yamanaka explained that the idea of reversing the fate of already specialized cells, then a ‘no-can-do’ in biology, came to him when studying the experiments that produced the first cloned frogs back in the 1970s, and Dolly the sheep in 1996. Both studies proved that the expression profile of the genome can be reprogrammed into a non-differentiated state if a differentiated cell nucleus is transplanted into an enucleated ovum.
“From their work I learned that we should be able to convert somatic cells back into their embryonic state. That is what inspired me to start my project,” he said.
Four genes, four transcription factors
Dr. Yamanaka hypothesized that if he could identify genes that are important for ES cells, he could then transfer those genes to a differentiated somatic cell — for example, an adult skin cell — and revert it back into its embryonic state.
Using an open database, his team identified 24 candidate genes expressed specifically in ES cells. After carrying out numerous experiments on such genes, he finally succeeded in generating iPS cells with the capacity for self-renewal and pluripotency similar to that of ES cells by introducing four genes (Oct3/4, Sox2, Klf4, c-Myc) into mice dermal fibroblast cells.
No one could have foreseen that such a limited number of factors were responsible for this complicated process.
The resultant iPS cells proved to have both the capacity for self-renewal and pluripotency, and his achievement was published in the science journal Cell in August 2006. Soon after in 2007, his team was able to generate iPS cells from human adult fibroblasts, so quickly that even he was surprised.
“At first I thought it would be very difficult, it would take 20 years, 30 years or more. But it took less than 10 years. We were pleased but at the same time surprised by our own results,” he said.
Can we reverse aging?
Dr. Yamanaka’s iPS cell technology is expected to aid in drug discovery and drug toxicity testing.
“Many people are trying to identify new drugs by using iPS cells from patients, so we hope that we will see some new treatments within a few years from now,” he said.
Additionally, iPS cells may someday provide insights into treatments for diseases that are currently considered incurable like type I diabetes, Parkinson’s disease and osteoporosis, by giving scientists the tools to reprogram cells back to their embryonic state and observe them as they develop in the lab.
But safety is a number one concern in this situation, says Yamanaka. Hence, the use of reprogrammed cells in cell therapy – with cells that are the patient’s own ‘offspring’ and therefore do not cause immune rejection – will take longer to reach the clinic, he cautions.
“We really have to double check the safeness of iPS cells,” he says, “to be sure that they don’t produce cancer and other types of tumors.”
Awards and recognitions

In 2007, Dr. Yamanaka was recognized as a “Person Who Mattered” in the Time Person of the Year edition of Time Magazine. He was also nominated as a 2008 Time 100 Finalist.
In 2009, he shared the Albert Lasker Basic Medical Research Award for his work in reverting adult cells to an embryonic-like state, thus circumventing an approach in which embryos would be destroyed.
In June 2010, he was awarded the Kyoto Prize, Japan’s highest private award for global achievement. Consisting of academic honors, it also includes a 20-karat gold medal and a cash gift of 50 million yen (US$600,000).
In February 2011, he received the BBVA Foundation Frontiers of Knowledge Award in the Biomedicine category with a cash prize of 400,000 euros.
Just today, he received the 2011 Albany Medical Center Prize in Medicine and Biomedical Research, along with Elaine Fuchs of Rockefeller University in New York City and James A. Thomson of the University of Wisconsin-Madison School of Medicine and Public Health. They will share US$500,000, the largest award in medicine and science in the United States.
His full list of awards is shown here:
• 2007 Meyenburg Cancer Research Award
• 2008 Yamazaki-Teiichi Prize in Biological Science & Technology
• 2008 Robert Koch Prize
• 2008 Shaw Prize in Life Science & Medicine
• 2008 Sankyo Takamine Memorial Award
• 2009 Lewis S. Rosenstiel Award for Distinguished Work in Basic Medical Research
• 2009 Gairdner Foundation International Award
• 2009 Albert Lasker Award for Basic Medical Research
• 2010 March of Dimes Prize in Developmental Biology
• 2010 Kyoto Prize in Biotechnology and medical technology
• 2010 Balzan Prize in biology
• 2011 Wolf Prize in medicine
• 2011 BBVA Foundation Frontiers of Knowledge Award
• 2011 Albany Medical Center Prize in Medicine and Biomedical Research
Oct. 8, 2012 UPDATE: Dr. Yamanaka wins the 2012 Nobel Prize In Medicine.
——
Source: Albany Medical College, Fundación BBVA and Wikipedia.org.
Disclaimer: This article does not necessarily reflect the views of AsianScientist or its staff.